Clinical Pharmacology Adaptive Therapy Group
Professor Banerji's group aims to study re-wiring of signal transduction to understand and overcome mechanisms of drug resistance and, in addition, to understand exploit cancer evolution using pharmacological tools.
We are focused on the re-wiring of signal transduction using established cell lines to control mechanisms of drug resistance and comprehend the evolution of cancer.
Rewiring of signal transduction
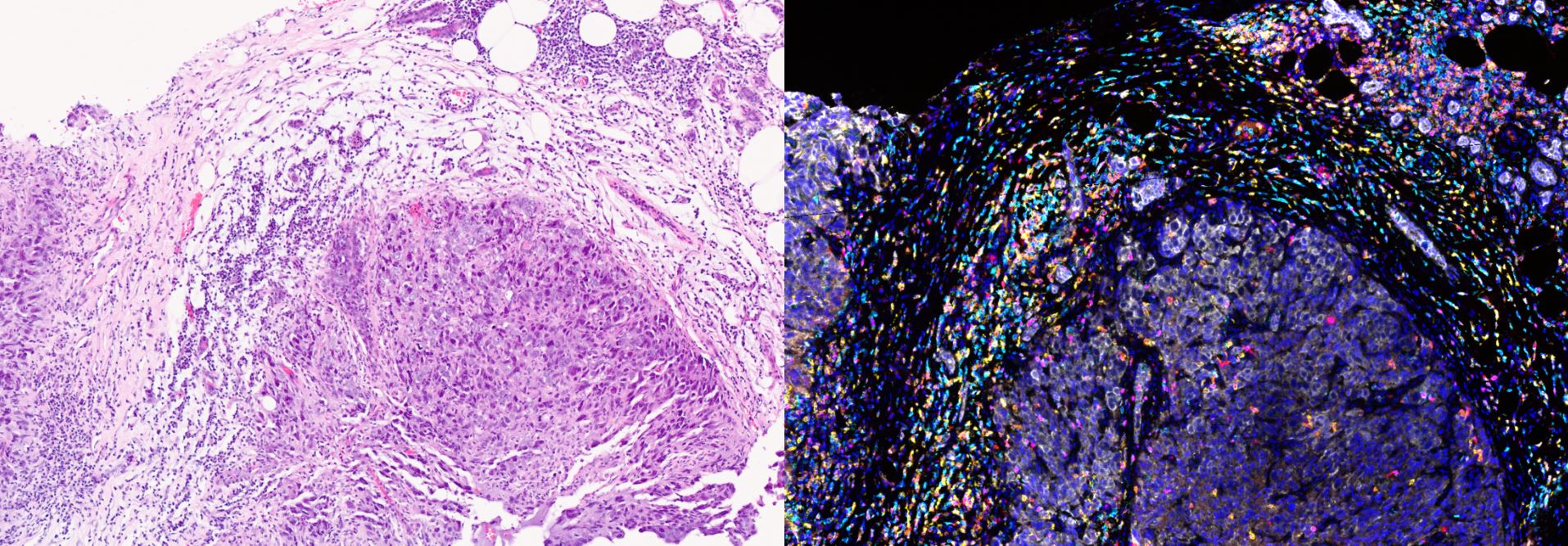
This group is working on the set-up and validation of a highly sensitive antibody-based assay on the Nanostring platform which will allow quantification of 50 - 100 phosphoproteins/proteins. It plans to digest tumour tissue obtained during surgery or from biopsies and expose them to a matrix of 20 - 30 anticancer drugs before obtaining phosphoproteomic data.
It also aims to use organoids and patient-derived xenografts to take this forward. The proteomic data will be modelled with the Computational Biology and Chemogenomics Team led by Professor Bissan Al-Lazikani. The group also supports PhD students studying the re-wiring of signal transduction in colon cancer and the role of stroma in influencing signal transduction.
The group also currently works with Professor Andrea Sottoriva at the Centre for Evolution and Cancer at the ICR to look at barcoding of cancer cells and the study of evolutionary trajectories of clones under experimental conditions. It is also studying mechanisms of resistance, collateral drug sensitivity and resistance of these emergent clones.
The primary area of this group is focused on the re-wiring of signal transduction using established cell lines as well as fresh cancer cells derived and isolated from patients and then exposing them to novel anticancer drugs. Pre- and post-proteomic profiling provides insights into mechanisms of drug resistance and how to overcome this with combination therapies. The initial work was carried out using cancer cells isolated from ascites and pleural effusions; the group in now developing expertise in organoid and patient-derived xenograft tissue.
The group develops its own antibody-based proteomic platforms and collaborates with teams involved in mass spectroscopic methods, led by Jyoti Choudhary. The group generates significant amounts of data and collaborates with the ICR's Computational Biology and Chemogenomics Team led by Professor Bissan Al-Lazikani to develop and decipher the data.
The secondary focus of this lab is the study of the pharmacological effects on cancer evolution in experimental models and methods to quantify this and herd cancer cells to a vulnerable state.
Professor Banerji works with Professor Andrea Sottoriva in the Centre for Evolution and Cancer at the ICR and plans to translate these concepts in to the clinic.
Vacancies at the ICR
Working at the ICR
Research Group Leader, ICR Clinical Trials and Statistics Unit (ICR-CTSU)
Department/Directorate Information: Division of Clinical Studies - Clinical Trials and Statistics Unit (ICR-CTSU) The ICR-CTSU is a Cancer Research UK-funded, internationally recognised methodologist led clinical trials unit, providing cancer-focused clinical trial research expertise. We lead pioneering, efficient, high-quality, and impactful trials across the phases. Our expertise ranges from experimental medicine early phase studies exploring biological efficacy to trials which may deliver widespread change to routine practice, underpinned by applied methodology to drive forward clinical trial innovation. See our clinical trials Role Summary The Group Leader will lead a component of ICR-CTSU’s portfolio of clinical trials research. The post holder will join an existing faculty and seek to further develop and grow the portfolio in line with ICR-CTSU’s overall strategy; taking responsibility for a number of ongoing trials as well as the development of new trials. There will be the opportunity to develop and grow a team including the potential for a Postdoctoral Training Fellow and/ or a Trial Manager. We seek an experienced statistician / biostatistician with a strong research interest in clinical trials methodology and a passion for direct involvement in the oversight and leadership of academic clinical trials. The successful candidate will work closely with the Director of ICR-CTSU to further enhance the Unit’s internationally recognised strength in clinical trial design, conduct and analysis. The post holder will be expected to make a substantial independent intellectual contribution to clinical trials projects and be proactive in leading and contributing to broad initiatives that enhance the overall effectiveness of ICR-CTSU. The appointee will contribute to the overall scientific life of the ICR including the newly established ICR/Royal Marsden Hospital’s Centre for Trials and Population Data Science, by providing mentorship to more junior colleagues and acting as an academic leader. We seek an individual who will work closely and collaboratively with other faculty/Group Leaders at the ICR and with international/national key opinion leaders to extend the breadth and depth of ICR-CTSU’s biologically rich clinical trials portfolio. In partnership with clinical opinion leaders, this individual will generate research funds to conduct and deliver clinical trials research at the international forefront. Presentation at national and international conferences, production of top-quality research outputs and substantial professional contribution to wider clinical trial network bodies are expected. Enthusiasm for team-based science in a collaborative interdisciplinary environment is essential. The appointment will be based on track record and the ability and willingness to engage in team science. The successful appointee will have access to ICR’s successful PhD training programme and core facilities. Key Requirements Higher degree (MSc or PhD) in medical statistics/biostatistics or an allied field (e.g. public health, epidemiology, data science) with relevant work experience Significant experience as a clinical trials, medical statistician or bio-statistician within the academic or commercial sector; a blend of both would be highly desirable A desire to apply existing and novel statistical methods to the requirements of a diverse range of statistical problems A broad understanding of cancer research Ability to lead a Clinical Trials Unit based research group As part of your online application, you will be required to upload your full CV which will pre-populate your application form, you will also be asked to attach the following documents and failure to do so will mean your application cannot be considered on this occasion: Lists of major publications, achievements, research grants and distinctions. A PDF of a maximum of five key publications, or other research outputs (e.g. patents) that best demonstrate previous productivity or a single document giving hyperlinks to these outputs. You must also complete the personal statement section of the application form in the format of a cover letter including the names and contact details of three academic referees Joining as a Group Leader, you will be given outstanding support to help you to continue to develop in your career. Along with a start-up package of funding, you will also have access to resources to establish your group, including support for recruiting key group members, such as PhD students and postdoctoral researchers. We encourage all applicants to access the job pack attached for more detailed information regarding this role. For an informal discussion regarding the role, please contact Professor Emma Hall ([email protected])
Postdoctoral Training Fellow - Biology (Cancer Therapeutics)
We have an opportunity for a talented and motivated Postdoctoral Training Fellow to join the Division of Cancer Therapeutics. The team has a role to accelerate the translation of ICR research into drug discovery programmes. The team delivers bespoke, hypothesis-driven data packages to support decision-making for launch of drug discovery projects. The teams dedicated resources, including biology, functional genomics, assay sciences, chemistry, and bioinformatics teams, will support joint project teams that combine the deep biological and therapeutic knowledge of ICR investigators with the validation and technical expertise of drug discovery scientists. Scientists in the team work closely with the Centre for Target Validation to validate and streamline robust, targets into drug discovery programmes in the ICR’s Centre for Cancer Drug Discovery or into collaborative programmes with external commercial therapy discovery partners. Also, working with the Centre for Protein Degradation Scientists exploit emerging technology and protein degradation tools for robust target validation experiments. The successful candidate will contribute to the development of new drug targets by expanding our understanding of target biology and drug discovery. Working in close collaboration with other groups within the ICR, the candidate will develop our understanding of the therapeutic relevance of specific targets using genetic and pharmacologic approaches. The successful candidate will be a part of a highly collaborative team that is embedded in the Centre for Cancer Drug Discovery. They will have the opportunity to direct their own research towards target identification, validation and drug discovery. About you Applicants must have a PhD in cell biology, cancer biology, genetics, or similar. Experience in drug discovery and modern cell and molecular biology techniques is essential. Experience with -omics level profiling and handling of large datasets is desired. Excellent organisational and communication skills are also required. Candidates who are nearing completion of their PhD may apply, but confirmation on awarded PhD is required within 6 months of employment. The ICR has a workforce agreement stating that Postdoctoral Training Fellows can only be employed for up to 7 years as PDTF at the ICR, providing total postdoctoral experience (including previous employment at this level elsewhere) does not exceed 7 years. For general information on Postdocs at The ICR, more information can be found here. Department/Directorate Information About our organisation We are one of the world’s most influential cancer research institutes with an outstanding record of achievement dating back more than 100 years. We are world leaders in identifying cancer genes, discovering cancer drugs and developing precision radiotherapy. Together with our hospital partner The Royal Marsden, we are rated in the top four centres for cancer research and treatment worldwide. As well as being a world-class institute, we are a college of the University of London. We came top in the league table of university research quality compiled from the Research Excellence Framework in 2014 and second in 2021. We have charitable status and rely on support from partner organisations, charities, donors and the general public. We have more than 1000 staff and postgraduate students across three sites – in Chelsea and Sutton. About our Centre The Centre for Cancer Drug Discovery (CCDD), within the Division of Cancer Therapeutics, is a multidisciplinary 'bench to bedside' centre, comprising around 160 staff dedicated to the discovery and development of novel therapeutics for the treatment of cancer. The CCDD’s exciting goal is to discover high quality drug candidates for validated biological targets and to progress these candidates to clinical trial. All the scientific disciplines are in place to make this possible. Our world-class biologists, chemists and drug metabolism specialists work together focusing on new molecular targets emerging from human genome and ground-breaking cell biology research. This is an exciting and fast-moving area of cancer research and offers the opportunity to work within a multi-disciplinary environment using state-of-the-art techniques and equipment. Group information Our group focus is to build robust target validation data packages against potential cancer targets identified by the ICR's researchers across Divisions. This is to increase confidence in the launch of drug discovery projects against those targets. Our scientists collaborate with ICR researchers to expand on the target biology validation and build a strong therapeutic hypothesis for a target. Tractability of targets is assessed and the most appropriate therapeutic mode of action for a target will be defined. Research at the Centre for Target Validation is focused on applying best practice in target validation and implementing innovative technology to robustly validate targets for drug discovery. Scientists in the CTV will form collaborations with ICR researchers who have identified potential therapeutic targets to: Provide access to resources and core facilities to answer hypothesis-testing questions Generate the key decision-making data required for target to enter drug discovery The Centre for Target Validation therefore links the Centre for Cancer Drug Discovery within the Division of Cancer Therapeutics with partner Divisions that identify potential drug targets. The Centre is also in collaboration with the Division of Breast Cancer Research to validate targets relevant in breast cancer in the context of the Breast Cancer Now (BCN) Toby Robins Research Centre. The CTV is supported and funded by the ICR as part of the ICR research strategy: Defeating Cancer to transform the lives of cancer patients. Group Leader: Joanna Loizou What we offer A dynamic and supportive research environment Access to state-of-the-art facilities and professional development opportunities Collaboration with leading researchers in the field Competitive salary and pension We encourage all applicants to access the job pack attached for more detailed information regarding this role. For an informal discussion regarding the role, please contact Joanna Loizou via [email protected].
Employee Story
Dr Fatemeh Ahmadi Moughari is a bioinformatician working in the Functional Genomics Team, led by Rachael Natrajan, and the Bioinformatics Team, led by Syed Haider. She joined the ICR from her home country of Iran where she completed her PhD in anti-cancer drug response at Shahid Beheshti University.
"I really enjoy the multidisciplinary atmosphere here and it’s the thing that stood out when I first joined the ICR."
Industrial partnership opportunities with this group
Opportunity: A potent, orally bioavailable clinical-stage inhibitor of MPS1 with potential as a treatment for a range of cancer types including triple negative breast cancer
Commissioner: Swen Hoelder