Some cancers, as I’ve blogged before, can be cured without medical help at all.
These cases offer a tantalising reminder of cancer’s fallibility – a glimpse of a possible future where cancer’s incredible complexity could be fully understood, and new treatments will control it by taking advantage of inbuilt weaknesses.
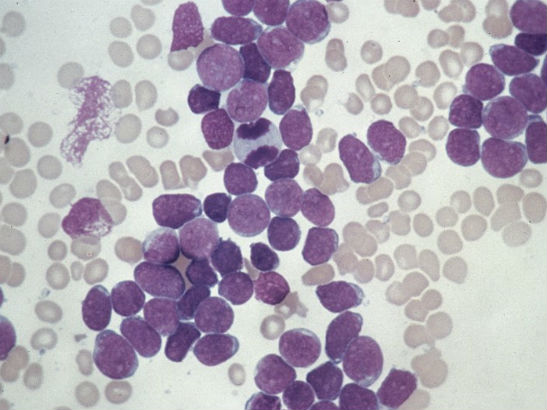
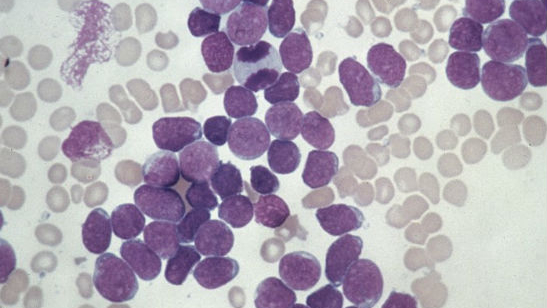
One ‘self-curing’ cancer is called transient amplifying leukaemia. Rare in the general population, it is very common in infants with Down syndrome – developing in around a third of children. The association occurs because the genetic cause of Down syndrome can also affect the action of the gene, called GATA1, which causes this type of leukaemia.
Usually there is no treatment required, because in most cases it simply resolves within a year. After an expansion in the number of cancerous stem cells before and after birth, they gradually die out, although in a few cases leukaemic stem cells will acquire additional mutations that lead to a more aggressive cancer, called acute megakaryoblastic leukemia (AMKL).
The question that has intrigued cancer researchers is why a population of cancerous stem cells – each equipped with the cellular machinery to make millions of copies of itself – spontaneously disappears?
A fascinating new article in Stem Cells by a team including Dr Benjamin Werner, from the ICR’s Centre for Evolution and Cancer, gives a possible explanation. And it could also help explain why childhood and adult cancers are often so different from each other.
Evolution
Dr Werner and colleagues developed a mathematical model to explain how a cancerous cell might have an evolutionary edge over its non-cancerous rivals early in life – but as the body’s development slows down after early childhood, this advantage could disappear.
We know that how a cancer develops depends on the ‘environmental’ conditions it lives in, as well as the genetic errors it accumulates. Just as an animal needs food, water, and shelter to live, cancers will only grow and spread if the environment is right – and subtle changes to the environment can have big effects.
A genetic error that might bring an advantage early in life – when cells throughout the body are growing and dividing rapidly – could cease to be an advantage later on when there is less growth and change in the body, the researchers’ model predicts.
Transient cancer
The researchers used their new model to look at transient leukaemia, and predict what might happen if a cancerous blood stem cell – the type of cell which causes leukaemia – had an early advantage which eventually fell away. The model predicted what happens in the disease, with an early expansion of cancerous cells building to make up around half of the total number of blood stem cells – and then gradually dying off to near-extinction later in life.
The evolutionary trajectory of this type of transient leukaemia is very similar between infants, which suggest a context-dependent fitness of the leukaemic stem cells that is closely tied to the natural regulatory mechanism of the hematopoietic system. Here the mathematical model offered an explanation.
Blood stem cells have an important role in the body: it is their job to populate the blood with the right balance of ‘worker’ blood cells. While they need to divide to create copies of themselves that can go onto create more blood cells in future, they also need to create a range of blood cells that have many different functions.
When blood stem cells divide into two daughter cells, they have three possible options. They can make two identical copies of themselves; they can make one exact copy, and a copy that is different to the original; or they can make two new different cells. In normal circumstances this process is finely balanced, meeting the need to produce all the cells of the blood, while also maintaining a population of stem cells to keep this production going all through life.
The researchers’ theory is that in transient leukaemia, a genetic mutation will give a cancerous cell the edge over normal stem cells by skewing this process towards the production of more identical copies than the norm – what they call an increased tendency towards ‘symmetric self-renewal’.
Cancerous mutations can then spread quickly through the population of stem cells because a cell which carries it will make more copies of itself than the average.
Changing the balance
But what shifts the balance back against the cancerous mutation?
After birth, overall development happens very quickly, with higher than normal levels of cell division – but gradually, it slows down. There are natural failsafe mechanisms built into the cells’ environment that cause an overall slow-down in stem cell production, nudging them to divide into new cell types rather than copy themselves. Naturally, the number of healthy stem cells in the body must be finite, as humans usually don’t grow indefinitely.
Eventually, the researchers’ theory says, this natural process of shifting the balance between self-renewal and differentiation will win out, and even the cancerous stem cells will be more likely to make new worker blood cells rather than copies of themselves. These cells won’t persist in the blood system forever, because they don’t have the capacity for unregulated growth their stem cell parents had.
If the cancer stem cells are more likely than not to divide into new cells they will gradually decline in number. The environmental conditions have changed, they are no longer at an advantage and gradually die out.
The phenomenon of shifting the advantage – changing ‘fitness’ for cancerous cells in a Darwinian sense, which is a well-studied mechanism in ecology – could happen in many cancer types, in incredibly complex ways. In their article, Dr Werner and colleagues argue that the shifting developmental landscape could explain the differences between adult and childhood cancers. Given they develop in such different environments, it makes sense that they would be quite different diseases.
The big question that this study poses – a question which drives researchers in our Centre for Evolution and Cancer – is how might we develop new treatments that shift the evolutionary advantage away from cancer cells?
As well as developing treatments that go after the players – the cancer cells themselves – could we develop new types of treatments that change the rules of the game, removing their advantage?
Only time will tell, but studies like this may provide the building blocks of knowledge to begin to answer these important questions.