
Photo: Jan Chlebik/the ICR
New discoveries in how we read DNA have helped scientists make huge strides in recent years towards identifying possible targets for new cancer drugs.
Modern techniques can process millions of base pairs of genetic code faster and cheaper than ever. Researchers can now read a cancer’s entire complement of DNA to identify genes that drive how tumours develop, known as oncogenes.
These new technologies greatly benefit research into new treatments for advanced prostate cancer, and the next decade holds much promise for improving our understanding of this disease to deliver better treatments.
This is vital because although prostate cancer is often treatable when caught early, once it has begun to spread round the body it typically will stop responding to conventional treatments.
The problem of resistance
Prostate cancer is the most common male cancer in the UK; every year, 47,000 men are diagnosed and 10,800 die from the disease. Normally, these lethal cases of prostate cancer are ones where the disease spreads to other organs — known as metastasis — and continues to worsen despite treatment.
Patients are initially treated by blocking the effects of hormones called androgens. In healthy men, androgens are involved in helping the body develop male characteristics, but prostate tumours also often come to rely on the hormones as a way of promoting their growth.
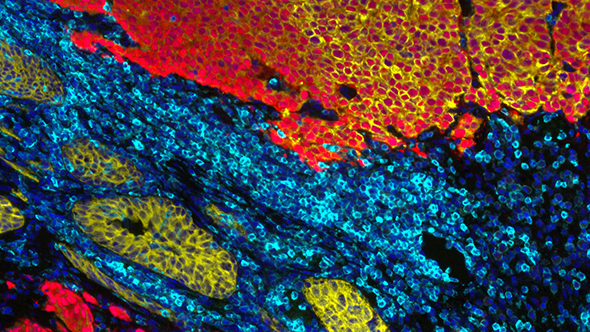
Immunofluorescence staining showing extension of prostatic adenocarcinoma into the bladder wall in a castration resistant prostate cancer patient (image: Mateus Crespo/Professor Johann de Bono, the ICR).
Androgen deprivation therapy remains the first line of treatment for advanced prostate cancer that has metastasised, either by using hormone treatments to block androgens, or very occasionally by removing the source of the hormone, the testicles.
Yet resistance to treatment often develops within two to three years, at which point it is known as castration-resistant prostate cancer. Prognosis is poor.
So the race is on to find new ways to treat patients whose initial therapy will fail.
Now, researchers at The Institute of Cancer Research and The Royal Marsden have published a far-reaching review in the journal Nature Reviews Drug Discovery of current progress and future trends in research into advanced prostate cancer.
Mapping genomes
In the paper, Professor Johann de Bono, Regius Professor of Cancer Research at the ICR and a consultant at The Royal Marsden NHS Foundation Trust, and colleagues say that recent mapping of the 'genomic landscape' of prostate cancer has helped us learn more about the biology of these tumours.
For instance, about nine in 10 prostate cancers possess genetic flaws that could be exploited in the clinic.
By analysing these genetic flaws, scientists believe they've found many novel potential targets for treatment. A range of studies and trials are under way to test whether these targets can affect tumour progress and patient outcomes.
The review’s authors list 15 potential new therapeutic targets for advanced prostate cancer, highlighting four as the most promising.

Professor Johann de Bono was recently named Regius Professor of Cancer Research at the ICR (photo: Jan Chlebik/the ICR)
The first of these targets are the androgen receptors, which sit on the surface of cells and bind to circulating androgen hormones.
Research shows two-thirds of patients have errors in the genes for these receptors. There is now renewed interest in deciphering how resistance to androgen deprivation therapy, or ADT, occurs, and numerous studies are under way to find new therapies to overcome it.
Targeting genetic flaws
Similarly, a set of chemical messengers called the PI3K-AKT signalling pathway is also showing promise. Recent research suggests targeting the body's immune system may provide an opportunity to treat cancers with errors in this pathway.
Recent studies have also identified a potential to target errors in another pathway, called Wnt signalling, in a subset of cancers that are not dependent on androgen. Early clinical trials are under way to determine whether this is a viable target for drug therapy.
In addition, researchers are investigating defects in the genetic machinery that repairs our DNA when errors occur.
Errors in genes involved in a biological process that repairs potentially harmful breaks in DNA — known as 'homologous recombination' — occur in up to a third of castration-resistant prostate cancers. And recent research at the ICR suggests nearly one in eight men (12%) with advanced prostate cancer have inherited mutations in one of several genes involved in DNA repair.
These defects can be targeted by drugs designed to kill cells, including so-called 'platinum drugs' and 'PARP inhibitors'. Learning how to identify which cancers have these specific defects could help scientists develop ways of predicting how a patient will respond to treatment.
Discovering what works for different patients
Drug development research in this field is fast-moving. Phase III trials testing a variety of chemical agents are now showing a survival benefit to these patients.
However, the ICR and Royal Marsden researchers say there is an ‘urgent need’ to determine the best order in which these new therapies should be administered for maximum benefit, depending on the patient’s disease characteristics.
This, they say, can be guided by improving how we classify different types of the disease on a molecular basis. But the lack of biomarkers — biological indicators of the current medical state — for predicting outcomes makes this difficult.
A further complication is that types of prostate cancer tumours are actually usually variable, or ‘heterogeneous’, in their make-up, both between different people and even within the same person's cancer.
Precision medicine now allows us to sub-divide people’s cancer into distinct subgroups based on the type of genetic errors present in their tumours, and arrange treatment accordingly. By studying how well these treatments work in different patients, and analysing their tumours at the molecular level, scientists are adding to a growing list of potential predictive biomarkers.
Targeting the immune response
Researchers are also exploring the potential to harness the immune system to fight advanced prostate cancer.
Our immune system hunts and destroys pathogenic agents by identifying small proteins on their surface. But some cancers can hide from the immune system by halting production of these proteins and by producing others that block an immune response.
Teaching the immune system to recognise the cancerous cells once more can overcome these avoidance mechanisms and allow it to attack. One such ‘immunotherapy’, called sipuleucel-T, is now approved for prostate cancer by the US Food and Drug Administration after being shown to improve overall survival by four months versus placebo.
A white blood cell of the immune system (photo: Wellcome Images)
And soon, progress in immunotherapies to treat advanced melanoma and non-small-cell lung cancer may reach patients with castration-resistant prostate cancer: several are under investigation for their potential as a therapy for this cancer.
On the horizon
Our researchers describe this as an exciting time for the management of prostate cancer, but they also outline big challenges that we must overcome.
We need to decide which drivers of advanced prostate cancers should be prioritised by researchers and clinicians. Separating out patients into groups based on their genetic profile is key to successful drug development, they say. And working out how the cancers will attempt to evade treatment will help to tailor the order of drugs given to a patient to the best effect.
In future, they add, we may even be able to track molecular changes in circulating tumour cells — potentially used as markers of the cancer state — in real time as cancer treatments are delivered.
We may also use the next generation of technologies to analyse how tumours evolve during treatment, and track the evolution of groups of cells that may be developing resistance.
In practical terms, future trials of potential drug candidates must also reflect these developments, and be ready to assess how the variation in tumours between and even within patients affects outcomes, the researchers explain. Trials will also need to test potential new biomarkers to aid precision medicine, and overcome ongoing issues with high trial drop-out rates.
Nevertheless, the outlook is optimistic. Groundbreaking new genomic technologies have arrived in recent years. Several effective new drugs are now approved, with many more under evaluation. And we’ve made significant progress in understanding more about drug resistance.
All these offer hope for further advances in patient care and, ultimately, survival.


