News and features
Read the latest news and features about our world-leading research, discoveries, fundraising and philanthropy. If you want to keep updated on our news, you can follow us on social media or sign up for our Search newsletter.
If you’re a journalist and want to find out more, you can contact our media relations team.

Scientists discover how to remove skin cancer’s protective armour and stop it spreading
Scientists have uncovered a protein that acts like a ‘suit of armour’ for cancer cells, shielding them from hostile environments and allowing one of the deadliest forms of skin cancer to spread through the body.
-is-malignancy-of-the-esophagus-cancer-of-the-esophagus-ct-with-contrast.jpg?sfvrsn=70ef5f67_1)
New study reveals how oesophageal cancer adapts to treatment
Researchers have tracked how the most common form of oesophageal cancer and its immune environment change during a standard form of treatment – offering vital clues that could shape future therapies, so they work for longer.
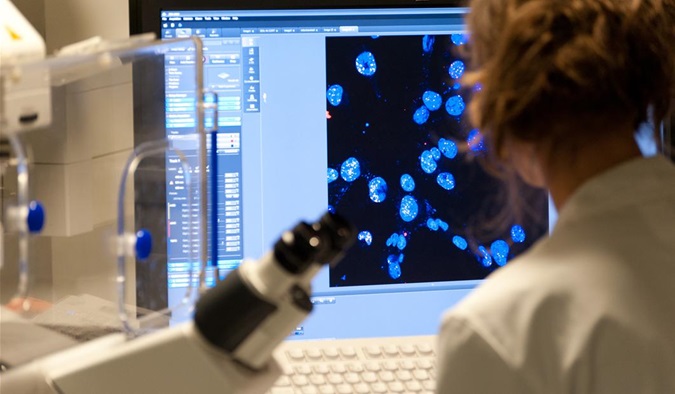
Newly discovered role of enzyme could explain link between diabetes and cancer
Cell biologists have made a significant advance, uncovering the key role of an enzyme in determining the physical structure and behaviour of cancer cells, including how they spread.

ICR researcher awarded prestigious lectureship for practice-changing breast cancer research
Dr Maggie Cheang, a breast cancer researcher at The Institute of Cancer Research, London, has been awarded the prestigious CL Oakley lectureship.

Scientists find new way to predict how bowel cancer drugs will stop working
Scientists from The Institute of Cancer Research, London, have developed a tool that can predict how bowel cancer adapts to treatment – helping researchers to design new personalised drugs that will keep patients living well for longer.

ICR scientist wins highly regarded award for early-career biomedical researchers
Dr Stephen John Sammut, a Clinician Scientist at The Institute of Cancer Research, London, has been awarded one of the eight 2025 research prizes from the Lister Institute of Preventive Medicine.
Scientists uncover clues behind drug resistance in bowel cancer
Scientists have discovered clues which may explain why some treatments stop working for people with bowel cancer – causing around 16,800 deaths in the UK every year.
The study, led by researchers at The Institute of Cancer Research, London, could help clinicians make better use of current treatments and develop more targeted therapies for bowel cancer in the future.
Thousands of blood cancer patients could benefit from new drug combination
Thousands of patients with a common type of blood cancer could benefit from a new drug combination, while others could see their disease kept at bay for longer.
Silencing gene may combat formation of radiation-induced scar tissue, a new study reveals
A new study suggests that silencing CXCL12, a gene involved in tissue scarring and repair, could help reduce the formation of scar tissue that can caused by radiotherapy. Ultimately, the researchers hope this approach could improve treatment outcomes for breast cancer patients undergoing radiotherapy and reconstructive surgery.
_kevin-harrington-embed.jpg?sfvrsn=cb25ea76_1)
ASCO 2025: One-time cell therapy offers long-term survival hope for patients with advanced melanoma
A one-time immunotherapy treatment using a patient’s own immune cells has shown long-lasting benefit for people with advanced melanoma, a serious form of skin cancer, according to new five-year follow-up data from a pivotal clinical trial.

ASCO 2025: Next-generation breast cancer drug targets tumours before they have a chance to grow
A powerful new drug for advanced breast cancer can be used to treat emerging tumours, months before they have a chance to grow, helping to keep patients well for longer and delaying the need for later-line therapies including chemotherapy.
Results of a global study, funded by AstraZeneca and co-led by researchers at The Institute of Cancer Research, London, The Royal Marsden NHS Foundation Trust and Institut Curie, Paris, were presented at the American Society of Clinical Oncology (ASCO) annual meeting in Chicago on 1 June 2025.
.jpg?sfvrsn=5e059cab_3)
ASCO 2025: New therapy improves survival in advanced breast cancer and delays need for chemotherapy
A promising new therapy can help patients with aggressive advanced breast cancer live longer and delays the need for further chemotherapy, new research has shown.
Final results of the INAVO120 study, led by an international team of researchers including scientists at The Institute of Cancer Research, London, and The Royal Marsden NHS Foundation Trust, have demonstrated the potential of the new therapy combination for targeting PIK3CA-mutated hormone receptor positive (HR+), human epidermal growth factor receptor 2 negative (HER2-) breast cancer – a common form of the disease.