Cancer cells display many different characteristics to normal tissue. Research from a new ICR team leader could use cancer’s unique metabolism to diagnose and treat the disease.
I recently attended our annual Faculty Retreat where I heard about some of the amazing new research happening here at The Institute of Cancer Research in London.
The ICR’s Faculty Retreat is a chance for senior staff to get together and review what we’ve done and where we’re going, but it’s also an opportunity for our newest recruits to present their work to the rest of the ICR and discuss how it fits within our overall strategic direction.
Four of our new team leaders were presenting their work to the faculty, giving us a tantalising glimpse into some exciting new areas of research. There was one presentation that caught my eye in particular.
At the ICR we are known for our work studying the genetic mutations that lead to cancer and targeting them with precision treatments, but a new area of research for the ICR is in understanding how genetic changes lead to alterations in tumour metabolism, which in turn help to fuel cancer’s uncontrolled growth.
Dr George Poulogiannis is leader of the ICR’s Signalling and Cancer Metabolism Team, and he gave a faculty talk titled: “Exploiting cancer metabolism in the era of personalised medicine”, discussing how understanding tumour metabolism could help doctors to select better treatments for patients.
While many mutations are needed before tumours form, and vary from patient to patient, most cancer cells show signs of having abnormal metabolisms. Changes in their signalling and metabolic circuitry adapt cancer cells to conditions in the body where oxygen or nutrients may be limited, so they could present ways to target and treat the disease, such as the well known phenomena called the Warburg Effect.
Normally cells create energy through a process called glycolysis, where glucose from food is converted into a compound called pyruvate, but in cancer cells glycolysis can occur at up to 200 times faster than normal. Scientists can track tumours by recording this boosted rate of glycolysis, and Dr Poulogiannis thinks that other metabolic mechanisms could offer new ways to identify cancer earlier.
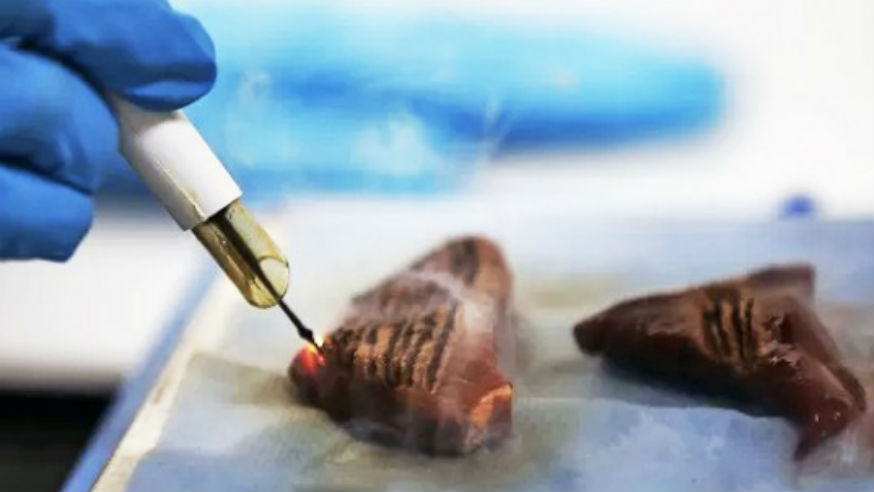
As part of his research at the ICR, Dr Poulogiannis will collaborate with colleagues at Imperial College London, Prof Zoltan Takats, Prof Jeremy Nicholson and Dr Kiril Veselkov to test their theories using their new iKnife technology, developed by Imperial scientist Professor Zoltan Takats.
The iKnife system consists of a mass spectrometer combined with electro-surgical tools. By measuring the chemical signatures in smoke given off when tissue is cauterised during routine surgery, the iKnife provides doctors with information about the tissue they are operating on.
The technology is still undergoing clinical trials, but the iKnife has already been used by Prof. Takats’ team to differentiate between healthy and cancerous tissue types. Dr Poulogiannis plans to join forces with Prof Takats and use this technology to explore the metabolic profiles of different cancer types.
Starting with DNA-sequenced breast cancer samples, their teams will build up a database of metabolic signatures and how they relate to underlying genetic changes. The eventual aim will be to classify tumours in real-time, and to understand how the molecular biology hidden behind certain metabolic signatures is associated with a patient’s prognosis or treatment selection.
The system requires more testing in patients but one day it could offer a cheap and instant way to profile tumours, helping doctors to select treatments tailored for individual patients without the need for DNA sequencing.