
The Aurora Borealis from a rooftop in Iceland (photo: Marco Ottobelli CC BY-SA 4.0)
You wouldn’t usually associate the bright, dancing display of the Aurora Borealis, the Northern Lights, with cells dividing.
A protein called Aurora got its name due to its localisation to the poles of the dividing cell, similar to the way the Aurora Borealis is observed at the poles of the earth.
Aurora kinases are essential for cell division and are often overactive or in high levels in cancer cells, which makes them an attractive target for cancer treatment. But although Aurora kinase small-molecule inhibitors have been around for a while now, it’s only recently that they have looked close to fulfilling their promise as potential cancer therapeutics.
I caught up with Dr Spiros Linardopoulos, Leader of the Drug Target Discovery Team at The Institute of Cancer Research, London, who has recently written a review with Dr Vassilios Bavetsias on the progress of Aurora kinase inhibitors.
What are Aurora kinases?
In humans, the Aurora kinase family consists of three members — Aurora-A, Aurora-B, and Aurora-C. Together they form an important group of enzymes that regulate many processes in cell division, including helping the dividing cell accurately dispense genetic information into two daughter cells.
If Aurora kinase signalling goes wrong, the cell’s chromosomes can be distributed unevenly, which is a dangerous state that can lead to cancer. And sure enough, Aurora kinase genes have been found to be overactive in a broad range of human tumours, including breast and ovarian cancers, gliomas, pancreatic cancers and haematological cancers.
Aurora kinase inhibitors were initially designed to target solid tumours including ovarian, breast, lung and bowel cancer. The clinical trials in these cancers were disappointing, but researchers did notice that the drugs seemed to work well in blood cancers such as leukaemia.
Response rates were more encouraging for Aurora kinase inhibitors that additionally targeted other factors that promote cancer. For instance, inhibiting another kinase called FLT3, which is known to drive certain blood cancers, gave promising anti-tumour activity in patients with acute myeloid leukaemia. Researchers are now exploring Aurora kinases that have dual inhibitor activity.
Double-pronged attack
A multidisciplinary drug discovery team led by Dr Linardopoulos has discovered a small-molecule dual inhibitor of Aurora and FLT3 kinases called CCT241736. Although FLT3 kinase has attracted a great interest in recent years as a target for acute myeloid leukaemia treatment, FLT3 inhibitors had limited success when used as single agents, because cancers tended to become resistant.
But in acute myeloid leukaemia cell lines that did not respond to FLT3 selective inhibitors, CCT241736 was able to overcome this resistance. CCT241736 has completed preclinical safety and toxicity studies and is poised to enter phase I clinical trials in patients with acute myeloid leukaemia.
Understanding the biology of Aurora kinases
Here at the ICR, one facet of our research strategy is to understand in detail the critical mechanisms used by cells to grow, divide and spread and how these processes go wrong in cancer.
As a continuation of the work focusing on Aurora kinases and their role in cancer, Dr Linardopoulos and his team have recently published a paper that sets out to establish what role Aurora-B and another mitotic kinase, namely MPS1, play in regulating the spindle assembly checkpoint — a pause in the cell cycle that allows the cell to check that all the chromosomes are properly attached to the mitotic spindle.
As the cell prepares for division, large protein complexes called kinetochores assemble to connect the chromosomes to the microtubule rope that forms the mitotic spindle. When kinetochores are not correctly attached to the spindle, they activate the spindle assembly checkpoint network, pausing cell division. Cell division can only continue when all kinetochores are stably and properly bound to microtubules, and the chromosomes are safely anchored.
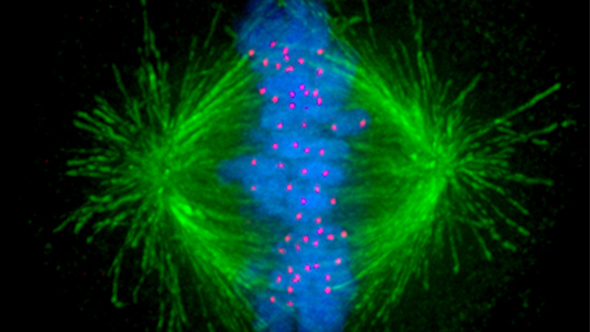
Image of the mitotic spindle in a human cell showing microtubules in green, chromosomes (DNA) in blue, and kinetochores in red.
In the Gurden et al. paper it was shown that Aurora-B has a dual role in maintaining the spindle assembly checkpoint signal. Firstly, it enhances the recruitment of MPS1 — an additional mitotic checkpoint protein — to the kinetochore, so that there is a rapid establishment of the spindle assembly checkpoint. But mainly, Aurora-B prevents the premature removal of spindle assembly checkpoint proteins, including MPS1, from unattached kinetochores.
Given the cross-regulation between Aurora-B and MPS1, researchers used breast cancer cells to test what happens when Aurora-B and MPS1 inhibitors were given together. Combining the drugs increased their potency, killing breast cancer cells by completely preventing the spindle assembly checkpoint from working. But more importantly, the drug combination sensitised breast cancer cell lines that otherwise did not respond to Aurora-B inhibitors alone. Dr Linardopoulos tells me that combining Aurora-B and MPS1 inhibitors may have great potential in the clinic as a cancer therapy, and his team will be exploring this further.
The future
So are Aurora kinases a new light in the sky for cancer treatments? Aurora kinase inhibitors are certainly emerging as promising targeted therapies for blood cancers, with interesting off-target effects that could be used to tackle the problem of resistance.
And our greater understanding of the biology of cancer is starting to open up potential new applications for this intriguing class of cancer treatment.