A team co-led by researchers at The Institute of Cancer Research, London, has uncovered how treatment-resistant cancer cells may drive poor outcomes for people with triple-negative breast cancer (TNBC), one of the most aggressive forms of the disease.
These cells already exist in tumours at the time of diagnosis and can survive chemotherapy, increasing the risk that the cancer will return or spread.
The study, published in Genome Medicine and funded by the charity Breast Cancer Now and others, has shown a new approach that could help doctors predict how well treatments will work for patients. The method also pinpoints important genes that could be targeted by new drugs. Together, this could help stop the cancer coming back in almost half of people with triple‑negative breast cancer (TNBC).
Addressing a major challenge
TNBC accounts for roughly 10–15 per cent of all breast cancers and most commonly affects Black women below the age of 40 who have inherited the BRCA2 gene. Unlike other breast cancer types, it lacks the key receptors that standard therapies rely on, such as oestrogen and progesterone, presenting fewer targeted treatment options.
Many patients receive neoadjuvant therapy, which is chemotherapy given before surgery to shrink tumours. While this treatment can be effective, around 45 per cent of patients are left with residual cancer cells in the breast (also known as drug-tolerant persister cells) presenting a much higher risk of recurrence.
Until now, it has remained unclear why these aggressive cells resist treatment. The Institute of Cancer Research (ICR) team, co-led by Dr Rachael Natrajan and Dr Syed Haider in the Breast Cancer Now Toby Robins Research Centre, within the Division of Breast Cancer Research, used a new biological technique called single-cell sequencing to investigate. Rather than studying tumours as a whole, this technique allowed the team to examine the activity of individual cancer cells in a tumour.
The researchers were able to analyse more than 129,000 individual TNBC cells, which were derived from laboratory models created from tumours that did not fully respond to chemotherapy and had residual disease.
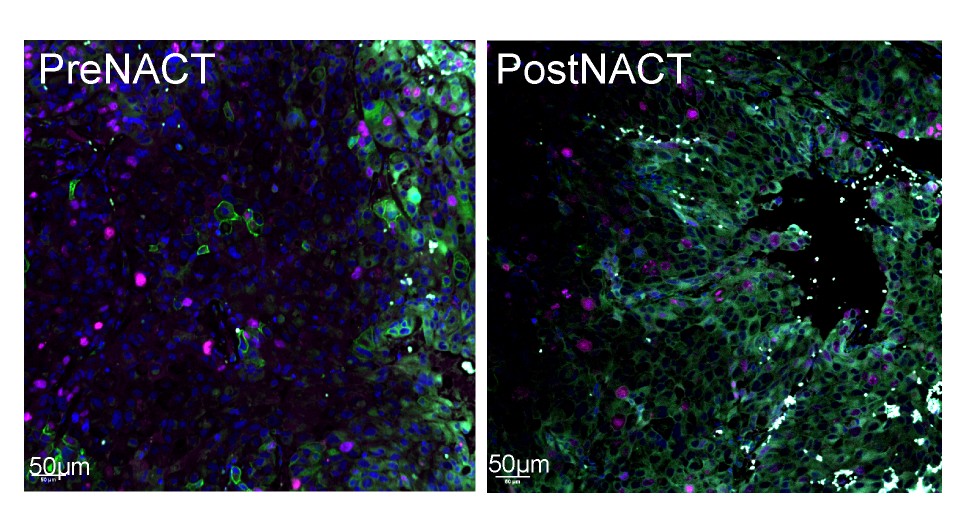
Image: The images above show a matched pair of patient derived TNBC tumours. PreNACT is where the sample was taken and engrafted in the mouse before the patient received any neoadjuvant chemotherapy and PostNACT is where the patient sample was taken after neoadjuvant therapy.
Cells poised to survive and spread
This analysis revealed that residual tumours often contain different types of cancer cells (cellular states) that are particularly resistant to treatment and linked to poorer long‑term outcomes for patients. Among these, the researchers identified two main groups of cells that share key features with drug‑tolerant persister cells. One group is adapted to survive in low‑oxygen (hypoxic) conditions, while the other shows a heightened activation of immune‑related signalling pathways.
The hypoxia-associated cells were found to be especially aggressive, carrying molecular features linked to long-term survival, treatment resistance and a greater ability to spread to other parts of the body.
Importantly, the study showed that these cells are not created when chemotherapy is administered but already exist within tumours. When exposed to chemotherapy, they can manipulate pathways to rapidly switch on survival tactics.
A new target to delay resistance
The team also identified several key regulators controlling these drug-tolerant persister cells, including a gene called KDM5B, which helps regulate gene expression programmes of drug tolerant cells and is activated under low oxygen conditions.
Dr Natrajan, Leader of the Functional Genomics Group at the ICR, said: “When we blocked KDM5B in our lab work, we were able to delay the emergence of these resistant cells after chemotherapy. While this is still early-stage research, it suggests that targeting this pathway could one day help prevent tumours from returning. With this new vulnerability to exploit, we can potentially stop these cells from taking hold after treatment and improve long-term outcomes for TNBC patients.”
What comes next?
Beyond identifying new drug targets, the findings could also help clinicians predict which patients are most at risk from the offset. By identifying the molecular signatures of drug-tolerant cells from tumour samples through single-cell sequencing, clinicians may also be able to identify patients who are unlikely to respond well to treatment.
Dr Haider, Leader of the Breast Cancer Data Science Group at the ICR, said: “This is a promising development for the field, but before our work can influence patient care, we must validate our findings within larger groups of patients, particularly those receiving chemotherapy combined with immunotherapy, which is now becoming the standard treatment for TNBC.
“Nonetheless, our study provides one of the most detailed pictures yet of why roughly half of TNBCs fail to respond to treatment, setting the groundwork for more personalised approaches for patients in the future.”
Image credit: ICR Science and Medical Imaging Competition 2022 Shortlist Image 3: A triple negative breast cancer cell on a lab-on-a-chip device by PhD students Melina Beykou (ICR) and Lewis Keeble (Imperial College, London)