Structural Biology of Cell Signalling Group
Dr Sebastian Guettler’s group is researching the ways in which certain enzymes, known as ADP-ribosyltransferases (ARTs), control cell function.
Research, projects and publications in this group
Our group utilises biochemistry, structural biology and cell biology to understand fundamental mechanisms of cell function. We hope to uncover novel potential therapeutic avenues in cancer.
We employ biochemistry, structural biology and cell biology to study the molecular mechanisms of Wnt/beta-catenin signalling and telomere homeostasis, with a particular focus on how poly(ADP-ribosyl)ation (PARylation) controls both processes. Besides understanding fundamental mechanisms of cell function, we aim to uncover novel potential therapeutic avenues in cancer.
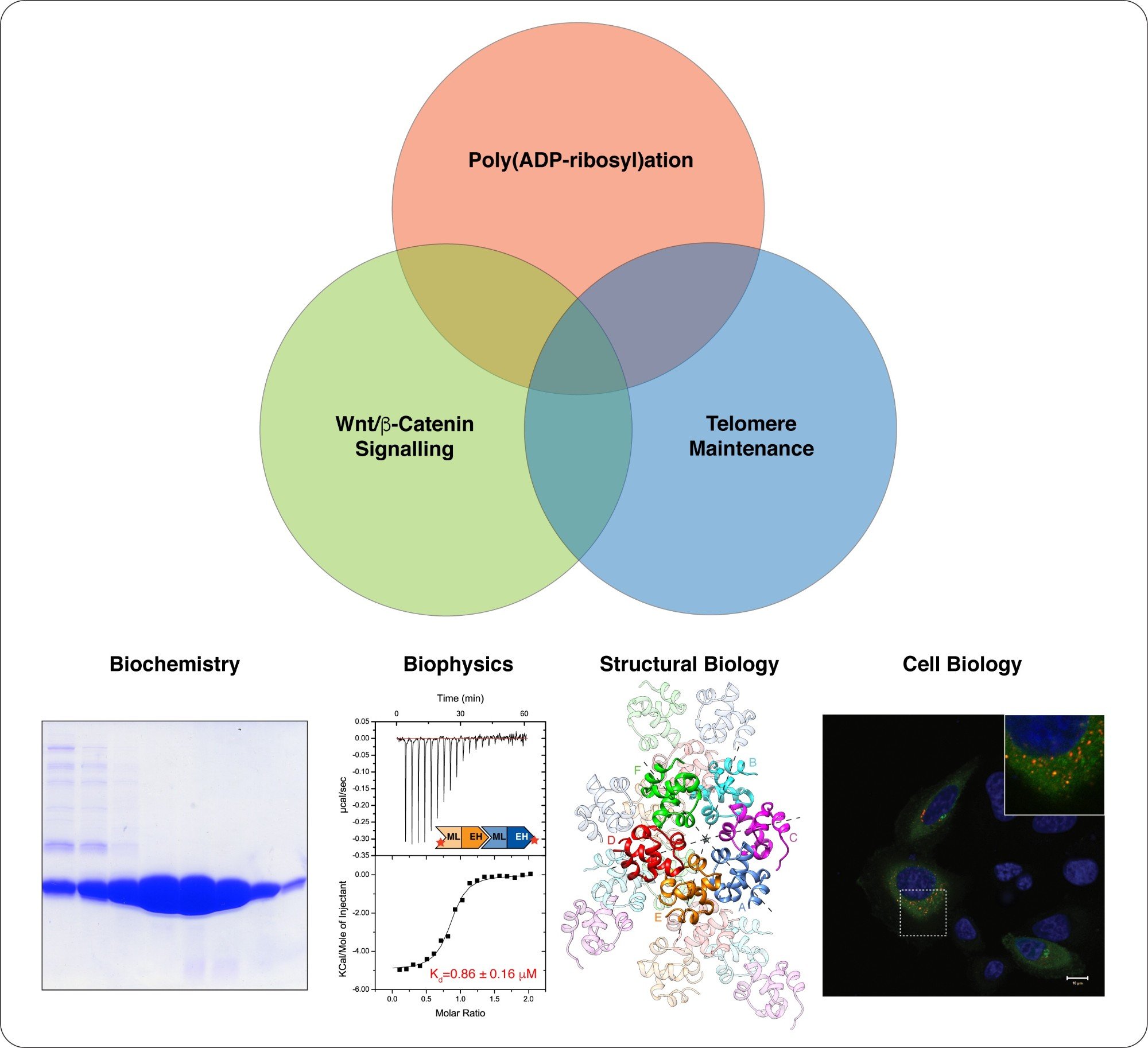
We take a multidisciplinary approach to study Wnt/beta-catenin signalling, telomere maintenance and their regulation by poly(ADP-ribosyl)ation. Structural biology is at the centre of our work. (Images modified from Mariotti et al., 2016)
A small number of key signalling pathways collaborate to confer stem-cell properties to cells, and the Wnt/beta-catenin pathway is a prototypic example for such a pathway. Wnt/beta-catenin signalling plays important roles in embryonic development and adult organ homeostasis. It is dysregulated in a number of different cancer types, most prominently in colorectal cancers, the vast majority of which bear mutations in components of the pathway.
At the same time, stem and most cancer cells rely on active telomerase to prevent erosion of their telomeres and maintain their unlimited replicative potential. Recent findings show that Wnt/beta-catenin signalling and telomere homeostasis are closely intertwined at multiple levels and form an integrated self-renewal programme, relevant to normal tissue regeneration, ageing and cancer.
The poly(ADP-ribose)polymerase (PARP) tankyrase both promotes Wnt/beta-catenin signalling and is essential for normal telomere extension in humans, thereby providing an important link between both processes.
Our overarching goal is to understand the precise molecular mechanisms that underlie Wnt/beta-catenin signalling, telomere maintenance and their control by poly(ADP-ribosyl)ation. We have a long-standing interest in deciphering the structural basis and molecular mechanisms of tankyrase function.
ADP-ribosylation is a post-translational modification carried out by ADP-ribosyltransferases (ARTs), which transfer ADP-ribose from NAD+ onto substrates. ADP-ribosylation controls many aspects of cell function, including DNA repair, cell division, telomere maintenance, chromatin dynamics, apoptosis and various signal transduction processes. Given their roles in DNA repair, telomere homeostasis and cancer-relevant signalling pathways, several ARTs are being explored as potential cancer therapy targets.
In humans, the family of intracellular ARTs encompasses 17 members with similar catalytic domains but greatly diverse non-catalytic accessory domains. Different catalytically active ARTs can either transfer a single unit of ADP-ribose or attach ADP-ribose processively, thereby constructing poly(ADP-ribose) (PAR) chains, which can be of varying length and structure. Enzymes in the latter group are known as poly(ADP-ribose)polymerases (PARPs). Compared to other types of post-translational modification, such as phosphorylation, PARylation remains understudied.
We take a particular interest in the PARP enzyme tankyrase, which fulfils a wide range of biological functions, many of which are relevant to cancer. The human genome encodes two highly similar tankyrase paralogues, TNKS and TNKS2. Both share a C-terminal catalytic PARP domain, a set of five N-terminal ankyrin repeat clusters (ARCs) responsible for substrate recruitment, and a polymerising sterile alpha motif (SAM) domain in between.
Our previous structure-function work has revealed the mechanisms of substrate recognition and polymerisation by tankyrase and shown that tankyrase can act as a scaffolding protein, independently of its catalytic function. We now aim to use both X-ray crystallography and cryo-electron microscopy to understand how tankyrase’s various domains act together. Moreover, we work with chemists to develop novel approaches to modulate tankyrase function.
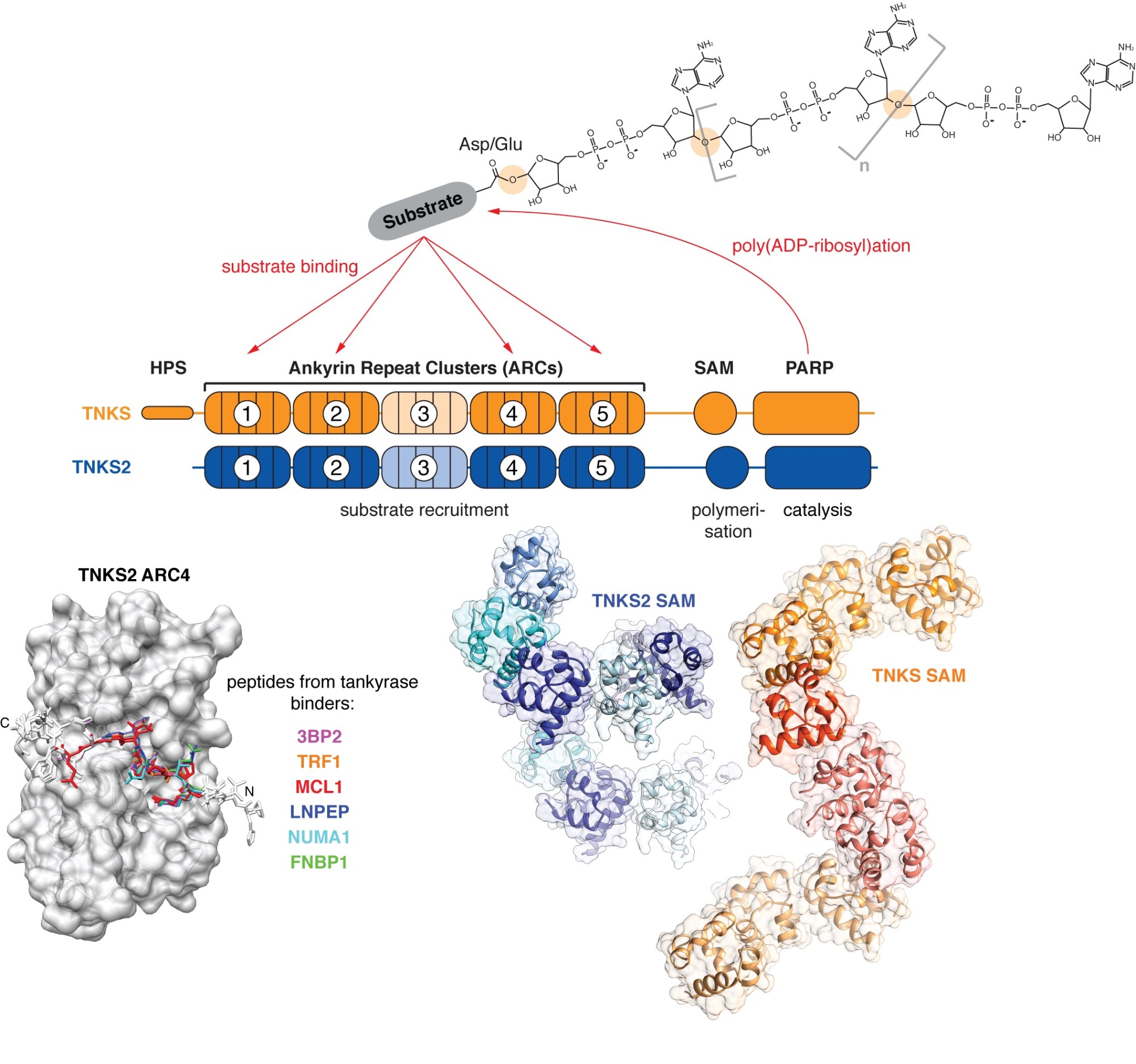
Tankyrase (TNKS, TNKS2) uses its ankyrin repeat clusters (ARCs) to recruit binding partners, many of which are also PARylated by tankyrase’s PARP domain. ARCs recognise degenerate peptide motifs found in many proteins. Our earlier work (Guettler et al., 2011) has revealed the substrate recognition mechanism and explained how the rare human disease Cherubism is caused. The sterile alpha motif (SAM) domain enables tankyrase polymerisation. We have revealed the mechanism of tankyrase polymerisation and shown that both ARCs and the SAM domain polymer fulfil essential scaffolding roles and are required for efficient substrate modification (Mariotti et al., 2016). (Images modified from Guettler et al., 2011; Mariotti et al., 2016 and Pollock et al., 2017)
Molecular mechanisms of Wnt/beta-catenin signalling, telomere maintenance and their regulation by poly(ADP-ribosyl)ation
In a series of projects, we take a reductionist approach to study how large macromolecular complexes coordinate Wnt/beta-catenin signalling and telomere length homeostasis and how they are controlled by tankyrase-dependent poly(ADP-ribosyl)ation. We combine biochemical assays with cryo-electron microscopy and X-ray crystallography to uncover the detailed mechanisms governing the functions of these complexes and their regulation.
Besides uncovering fundamental mechanisms underlying stem and cancer cell function, we endeavour to understand the molecular basis of disease mutations and open up new opportunities for pharmacological intervention.
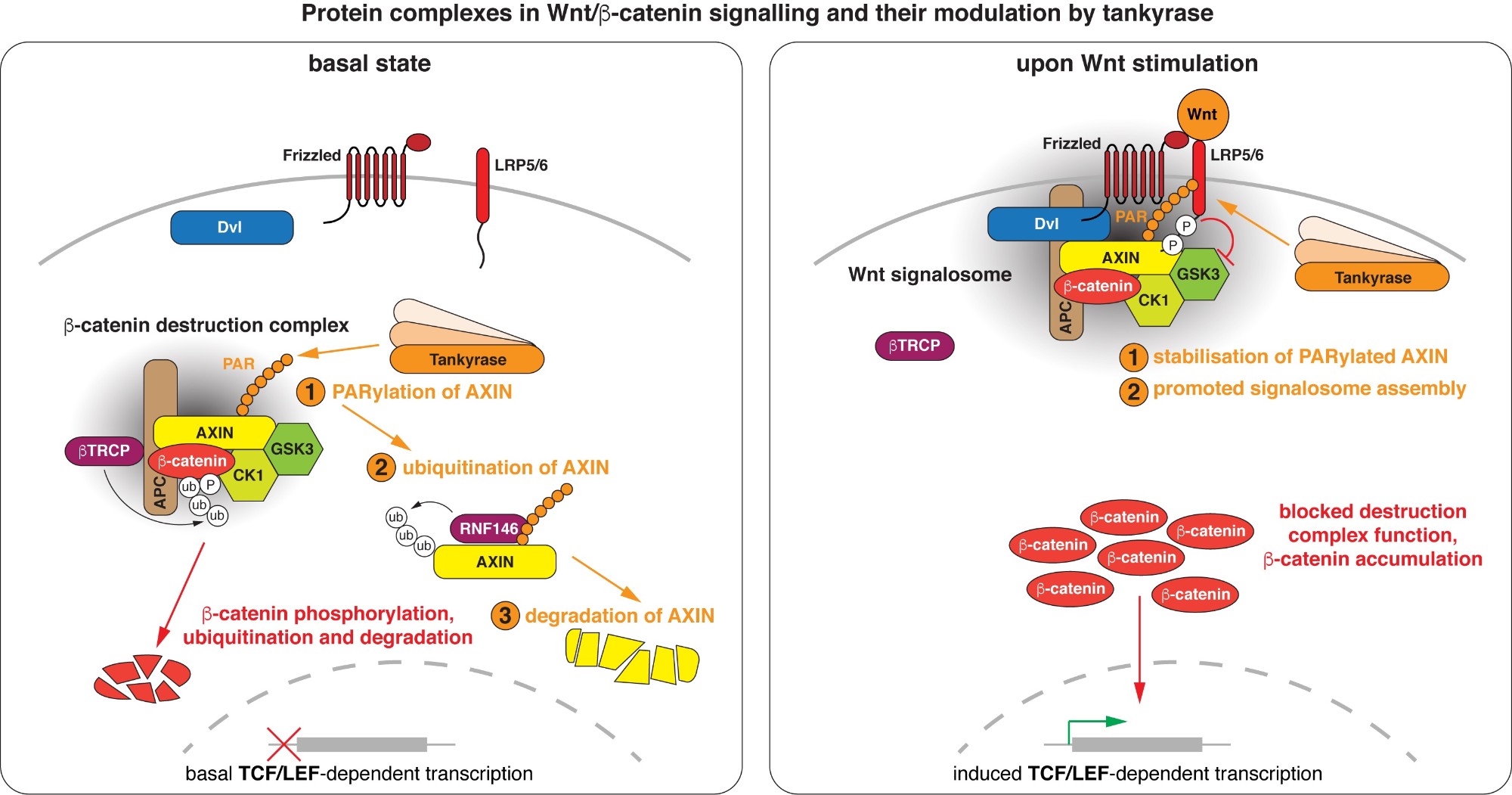
Wnt/beta-catenin signalling revolves around controlling the levels of the transcriptional co-activator beta-catenin. A multi-protein beta-catenin destruction complex captures cytoplasmic beta-catenin and limits its abundance by initiating its phosphorylation- and ubiquitination-dependent degradation. Notably, destruction complex function is impaired in the vast majority of colorectal cancer cases. Wnt stimulation remodels the destruction complex into a membrane-localised “Wnt signalosome” incapable of destabilising beta-catenin. Tankyrase controls the receptiveness of cells to incoming Wnt signals by PARylating AXIN, thereby destabilising the destruction complex or promoting Wnt signalosome formation. (Images modified from Mariotti et al., 2017)
Professor Sebastian Guettler
Deputy Head of Division:
Structural Biology of Cell Signalling.jpg?sfvrsn=4c485212_1)
Professor Sebastian Guettler is Deputy Head of the Division of Structural Biology. He studies the precise molecular mechanisms of signalling processes central to cancer stem cell function, with a particular interest in Wnt/β-catenin signalling, telomere length homeostasis and their regulation by ADP-ribosylation. He has a long-standing interest in understanding tankyrase, a poly-ADP-ribosyltransferase with roles in both Wnt/β-catenin signalling and telomere maintenance.
Researchers in this group
 .
.
Email: [email protected]
Location: Chelsea
I completed my MChem at the University of York in Chemistry, Medicinal and Biological Chemistry, with a year in industry, where I worked at UCB Pharma, in the Medicinal Chemistry department. My Masters project was on a drug discovery project in peptide therapeutics. After this, I completed a 10-week internship in organic chemistry at AstraZeneca. I then worked as a Junior Medicinal Chemist for a year, at Autifony Therapeutics, working in CNS drug discovery. For my PhD, an MRC iCASE-funded project, I aim to develop small molecules targeting the telomeric Shelterin complex, jointly in Sebastian Guettler's and Swen Hoelder's groups at the ICR, with Merck as our industry partner.
 .
.
Phone: +44 20 3437 6624
Email: [email protected]
I graduated from the University of Southampton, with an integrated Master’s in Biochemistry (MSc). For my bachelor’s project, I investigated the effects of reactive oxygen species and vitamin C on S100A9 aggregation in Alzheimer’s disease. For my Master’s project, I investigated the functional importance of Snap29 for mitosis in Drosophila. For my PhD, I study the molecular mechanisms of telomere maintenance.
 .
.
Email: [email protected]
Location: Chelsea
Oviya studied Biotechnology during her undergraduate degree at SRM University, India. She then completed her PhD in Professor Sara Sandin's lab at Nanyang Technological University, Singapore. Oviya joined the ICR as a postdoc in 2019 and is currently studying the function of tankyrase at telomeres.
 .
.
I completed my BSc degree at the University of West Indies (UWI), in Kingston Jamaica. I then pursued an MSc degree at the University College London (UCL), where I characterised a novel cause of Silver-Russell Syndrome in the lab of Professor Gudrun Moore. Subsequently, I ventured off to Zürich, where I joined the lab of Professor Ulrike Kutay at the Institute of Biochemistry, ETH Zürich, as a PhD student. My PhD research identified mammalian inner nuclear membrane proteins that majorly contribute to the establishment and the maintenance of 3D genome architecture, and the resulting biological consequences when this organisation is perturbed. I joined the Guettler Lab as a Postdoc in October 2025, and my work involves investigating the molecular mechanisms of tankyrase, particularly in Wnt/β-catenin signalling and telomere length homeostasis.
 .
.
Katy completed her MChem degree at Durham University in 2023, where her final-year research project focussed on the application of peptidomimetics to generate novel therapeutics for Alzheimer's disease. At the ICR, she's working on a collaborative project with Professor Sebastian Guettler and Professor Swen Hoelder, developing novel scaffolding inhibitors of tankyrase.
 .
.
Dr Vasundara Srinivasan obtained her PhD in Biochemistry from the University of Georgia, USA in the lab of Prof B.C. Wang. She next joined the lab of the Nobel Laureate Prof Hartmut Michel at the Max Planck Institute of Biophysics, Germany to work on membrane proteins, G-protein coupled receptors (GPCR) and transporters, focussing on mitochondrial ABC transporters in collaboration with Prof Roland Lill. As a project leader at the VIB, Brussels, Belgium, she used nanobodies as chaperones for the crystallisation of GPCRs. She headed the crystallisation facility at the University of Marburg before joining the lab of Prof Christian Betzel at DESY, Hamburg to work on the structure-based drug discovery of proteases involved in the replication cycle of SARS-CoV-2. She joined the lab of Prof Sebastian Guettler at the ICR in January 2025.
Professor Sebastian Guettler's group have written 29 publications
Most recent new publication 4/2025
See all their publications .
.
 .
.



