Video: "The work that we do now impacts generations of cancer patients in the future." Watch Professors Udai Banerji and Ian Collins talk about their work on AKT inhibitors.
AKT – which is also known as Protein Kinase B, or PKB – is an enzyme that plays a key role in a mechanism that many cancer cells rely on to survive, grow and resist treatment.
“AKT is an attractive target for the development of anti-cancer drugs,” says Professor Ian Collins, Head of Chemistry in the Cancer Therapeutics Unit at the ICR. “It’s one of the most important nodes in a signalling network that is commonly disrupted in many types of cancer, including breast, lung, ovarian and prostate cancers.”
Back in the early 2000s, identifying AKT inhibitors – molecules that block its action – had been hindered by the lack of protein structural information.
“The absence of structural knowledge made it difficult to design drug compounds that would block the activity of AKT,” explains Collins. “As random screening approaches also hadn’t generated many hit compounds, we needed that detailed information about the protein structure to move forward.”
Solving the structure
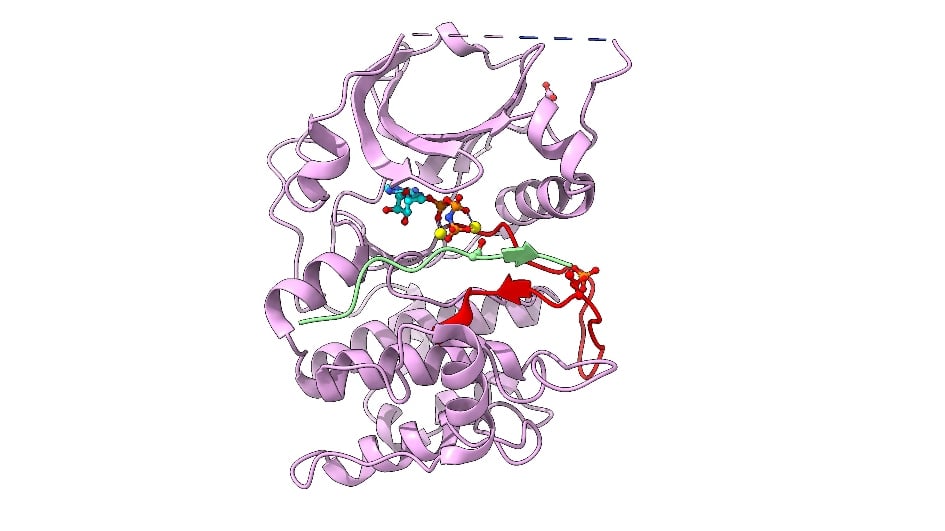
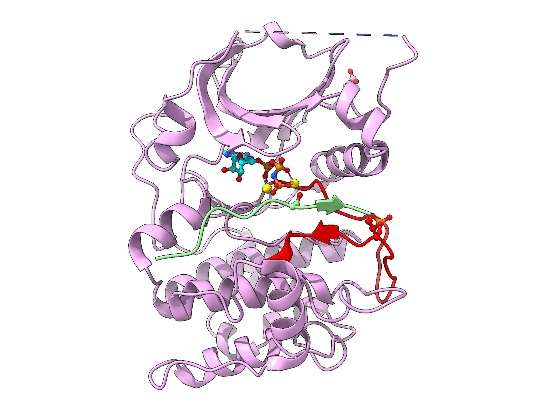
Image: Cartoon representation of AKT2 (PKB beta) kinase domain. Kinase domain in pink, activation segment in red, GSK3 substrate peptide in light green. The phosphorylated Thr309 residue (activation segment) and Asp474 (phosphoSer474 mimic), and ATP-Mn are shown. Credit: David Barford
A team of ICR scientists led by Professor David Barford set out to crack the 3D structure of AKT.
AKT is a member of the protein kinase family, which are enzymes that ‘switch on’ the activity of other proteins by phosphorylating – or adding a chemical group containing phosphorus – to specific amino acids, which are the building blocks of proteins.
But the activity of AKT itself is also switched on through phosphorylation by other protein kinases. In the early 2000s, scientists had established that the AKT protein is activated by two separate phosphorylation events at two different amino acid building blocks – one called threonine-309 (Thr309), and another called serine-474 (Ser474).
But one crucial piece of the puzzle was still missing.
“At the time, it wasn’t known which protein kinase phosphorylated Ser474,” says Barford. “So there was no way of generating activated AKT protein for structural studies.”
To circumvent this problem, the ICR researchers needed to invent a way to mimic the effects of AKT phosphorylation at Ser474. Painstakingly, they devised two different methods using innovative protein engineering.
“Our system circumvented the need to phosphorylate Ser474 to activate AKT,” says Barford. “That was a huge step forward, enabling us to generate large amounts of fully activated AKT for use in structural biochemical and functional studies.”
In 2002, the ICR researchers, in collaboration with a team at the Friedrich Miescher Institute in Switzerland, published the crystal structure of AKT and provided a molecular explanation for how AKT is switched on through phosphorylation at Ser474 and Thr309, and how it recognises its protein substrates. The team made the system for expressing and producing activated AKT widely available to companies and academic researchers, helping to accelerate drug discovery programmes around the world.
Searching for AKT inhibitors

Following these important discoveries on the structure and function of AKT, Professor Michelle Garrett and Professor Paul Workman kicked off a drug discovery programme at the ICR. Together, Professors Garrett, Workman and Barford – along with Professor Collins as the lead chemist– began the search for AKT inhibitors.
Their goal was to identify a small molecule that could fit snugly into an important site in the protein structure called the ‘ATP binding pocket’, which they hoped would block the entry of ATP – a source of phosphate – and inhibit the activity of AKT. In 2003, the ICR team began a collaboration with UK company Astex on this research programme.
The partnership between the ICR and Astex saw the use of several innovative approaches, including fragment-based discovery – which involved a combination of virtual compound screening and high-throughput crystallography.
“The strategy involved taking very small chemical compounds that are individually too small to have strong interactions with the protein, but are capable of fitting into lots of tiny crevices in the structure,” explains Collins. “Using x-ray crystallography, it’s possible to see these little fragments binding to the protein – and you can then use traditional synthetic chemistry to join them together into a larger molecule. It’s like you’re joining the dots within the structure to get the perfect shape to fit that site.”
The collaborative ICR-Astex drug discovery programme led to the rapid identification of compounds that blocked the activity of AKT and were suitable for further development.
“One of the biggest challenges we encountered was identifying compounds that were selective for binding to AKT, but not to other similar kinases – as this could lead to unwanted side effects,” says Collins. “We found a solution to this problem and we were surprised at how small changes to our compounds could make such a difference – it all came down to interacting with just one or two amino acids specific to the binding site of AKT.”
In 2005, a series of precursor drug compounds discovered by the ICR and Astex was licensed to AstraZeneca. In 2010, AstraZeneca announced its discovery of capivasertib (also called AZD5363) and began to develop the drug as a potential treatment for various types of cancer.
Image: Professor Ian Collins, Head of Chemistry in the Cancer Therapeutics Unit at the ICR
Entering the clinic
 The first-in-human trial of capivasertib was led by Professor Udai Banerji, Deputy Director of the Drug Development Unit at the ICR and The Royal Marsden NHS Foundation Trust. The phase I study involved 90 patients with a variety of cancer types treated at a range of centres across Europe and North America and found the drug was safe, allowing it to move to the next stage of development.
The first-in-human trial of capivasertib was led by Professor Udai Banerji, Deputy Director of the Drug Development Unit at the ICR and The Royal Marsden NHS Foundation Trust. The phase I study involved 90 patients with a variety of cancer types treated at a range of centres across Europe and North America and found the drug was safe, allowing it to move to the next stage of development.
The researchers also tested capivasertib in 54 women with breast, ovarian, cervical or womb cancer who had faults in a gene called PIK3CA, which helps to activate AKT – with the results providing early indications that it could shrink tumours in these patients.
“I remember seeing the first patients with PI3KCA mutations whose tumours had shrunk,” says Banerji. “That was extremely exciting.”
The researchers also carried out tests, or assays, on blood, hair and tumour samples, which were developed and run at the ICR. As well as providing evidence that the drug was able to block AKT, their results were pivotal in determining the dose and schedule to take forward into phase II trials.
“It’s only possible to look at what’s happening before and after treatment using tumour biopsies,” explains Banerji. “But developing assays on blood and hair samples meant we could profile if the drug was hitting the target at multiple timepoints – and this enabled us to work out that the drug should be given twice a day.”
The Pharmacological Audit Trail, developed by Professor Workman, played a crucial roles in getting the dose and schedule right to balance efficacy and tolerability in early trials and set the stage for later phases of development.
A subsequent clinical study, which also involved Professor Banerji, showed encouraging results for capivasertib in combination with fulvestrant, a current hormone treatment, for treating certain breast cancers with AKT mutations.
Following a successful phase II trial, and by now in 2020, the phase III CAPItello-291 trial was initiated. This trial aims to recruit 830 patients with advanced hormone-resistant breast cancer across 20 countries to examine the effects of capivasertib combined with fulvestrant.
If successful, the trial could establish the combination of the two drugs as a new breast cancer treatment regimen.
Image: Professor Udai Banerji, Deputy Director of the Drug Development Unit at the ICR and The Royal Marsden
Capivasertib and chemotherapy
Meanwhile, the BEECH study – led by Professor Nick Turner, Professor of Molecular Oncology at the ICR and Head of the Ralph Lauren Centre for Breast Cancer Research at The Royal Marsden – showed that capivasertib combined with chemotherapy could benefit women with a type of breast cancer that is particularly difficult to treat. The follow-on phase II PAKT study, which also involved Professor Turner, indicated the benefits of this combination for patients with advanced ‘triple-negative’ breast cancer.
As of December 2020, there have been at least 27 clinical trials of capivasertib, providing patients with experimental treatment opportunities. These include a phase III study to evaluate the drug in combination with abiraterone (a drug discovered at the ICR, and jointly developed with the Royal Marsden), which aims to enrol 1,000 patients with prostate cancer with faults in the PTEN gene (which switches off AKT).
And a phase I trial at the ICR and The Royal Marsden that evaluated capivasertib with another precision medicine, olaparib, showed this drug combination was safe, successfully hits its targets, and was effective against a variety of cancers, including those that had stopped responding to chemotherapy.
Several other AKT inhibitors have entered clinical studies including GDC-0068 (ipatasertib), GSK2110183 (afuresertib) and MK2206, which are being evaluated for treating different cancers including prostate, blood, stomach and bowel cancers in combination with other treatments.
For example, Professor Johann de Bono, Director of Drug Development at the ICR and The Royal Marsden, led a phase III trial of ipatasertib combined with abiraterone which showed this combination may offer potential benefits for treating men with advanced PTEN-deficient prostate cancers who currently have few treatment options.
Hope for patients
This work provides a compelling example of the ICR’s excellence at translating scientific discoveries about the biology of cancer cells into innovative new cancer treatments with real benefits for patients. Our researchers pioneered the idea of targeting AKT, revealed the 3D structure, and worked closely with industrial partners to drive the discovery and development of capivasertib, and with colleagues at The Royal Marsden to bring it to patients.
“It’s very exciting,” enthuses Collins. “It’s been very rewarding to see it come back to the ICR at various stages and be pushed along by other innovative contributions by ICR scientists, particularly at the clinical stage.”
Capivasertib has now entered three major phase III trials for hormone-resistant advanced breast cancer, triple-negative breast cancer and PTEN-deficient prostate cancers – which, if successful, could provide urgently needed new treatment options for patients with advanced disease that has become resistant to other treatments.
“It is a translational researcher’s dream to see a discovery move from the laboratory into something that can benefit patients,” says Banerji.
The REF is the UK’s system for the assessment of research quality and impact at its higher education institutions. To mark our outstanding success, we are celebrating some of the ICR research programmes that have been changing the lives of cancer patients.
Read more about REF2021