A four-drug combination therapy that includes the novel drug carfilzomib can slow the progression of the blood cancer myeloma in patients who have been newly diagnosed with the disease, a phase III study has shown.
Results from the Myeloma XI+ trial established that a combination therapy of carfilzomib along with the drugs, lenalidomide, dexamethasone and cyclophosphamide was well tolerated and can prolong the duration of first remission in patients with newly diagnosed myeloma.
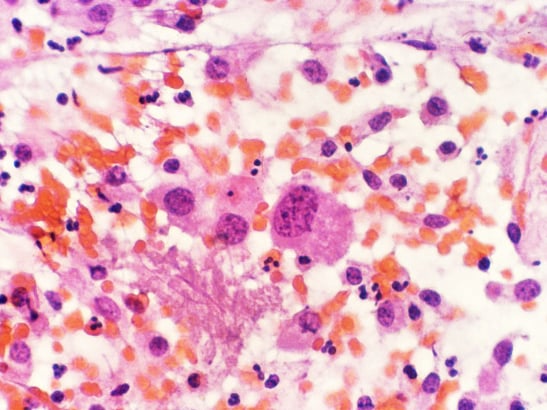
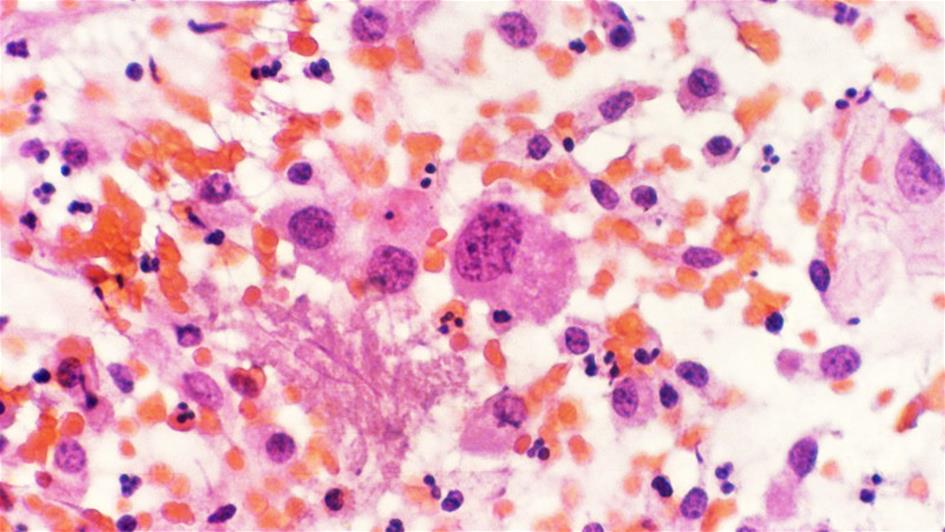
Myeloma develops from white blood cells in the bone marrow, called plasma cells, and accounts for 15 per cent of blood cancers. Around 5,700 people are diagnosed with myeloma each year in the UK.
A new analysis of the clinical trial results, led by researchers from The Institute of Cancer Research, London and our partner hospital The Royal Marsden NHS Foundation Trust, and Newcastle University, have been published in the journal PLOS Medicine.
They show that patients with the disease who received the combination therapy, denoted KRdc, prior to autologous stem cell transplant had a 37 per cent reduced risk of disease progression or death overall – compared with those receiving the triplet therapy without carfilzomib prior to their transplant.
The clinical trial overall – the first phase III study of carfilzomib combined with lenalidomide for newly diagnosed transplant eligible myeloma patients – was led by a team of myeloma investigators from 110 sites across the UK, including researchers from the Universities of Leeds and Newcastle.
It was largely funded by Cancer Research UK, with further funding from pharmaceutical companies Amgen – the manufacturer of carfilzomib under the brand name Kyprolis – Celgene Corporation, and Merck, Sharp and Dohme.
New diagnosis
Despite recent therapeutic advances in treating myeloma, many patients will still relapse, necessitating new therapies that target the disease earlier.
Carfilzomib is a member of a class of drugs called proteasome inhibitors. It has been shown to be effective against relapsed myeloma in combination with lenalidomide, with both drugs targeting cancerous myeloma cells directly using different mechanisms.
The Myeloma XI+ trial involved 1,056 patients with newly diagnosed myeloma, who were randomly assigned to receive either the quadruplet KRdc therapy or a control triplet option.
Three years after starting in the trial, 66 per cent of patients who received KRdc remained alive and progression free versus only 50 per cent in the control group.
In addition, 82 per cent of patients in the KRdc group achieved a very good partial response – defined as a 90 per cent of more reduction in measurable myeloma – compared with 59 per cent of patients in the control group.
Some multi-drug combinations can increase toxicity, which can make them unsuitable for clinical use. But the researchers found that the quadruplet KRdc therapy didn’t notably increase side effects.
Carfilzomib is known to be less likely than other proteasome inhibitor drugs to elicit peripheral neuropathy – a key side effect of several myeloma treatments that results from damage to nerves and can cause numbness and pain.
The study’s leading co-author Dr Charlotte Pawlyn, Team Leader in Myeloma Biology and Therapeutics at the ICR and Consultant in Haematology at The Royal Marsden, said:
“The results of this major, long-running clinical trial in patients with myeloma have established the benefit of carfilzomib in a quadruplet combination with lenalidomide, including in patients with newly diagnosed disease. We hope that these results now lead to the drug being approved for wider use on the NHS, as it is currently only available to UK patients in limited settings.”
Support our research today to unlock new combination treatments, so more people will survive cancer.
Let’s finish cancer, together.
Donate today